Andrei V. Alyokhin1*, Benjamin M. Rosenthal2, Donald C. Weber3, and Mitchell B. Baker4
1School of Biology and Ecology, University of Maine, 5722 Dering Hall, Orono, ME 04469, USA
2Animal Parasitic Diseases Laboratory, Agricultural Research Service, US Department of Agriculture, 10300 Baltimore Avenue, Beltsville, MD 20705, USA
3Invasive Insect Biocontrol and Behaviour Laboratory, Agricultural Research Service, US Department of Agriculture, 10300 Baltimore Avenue, Beltsville, MD 20705, USA
4Biology Department, Queens College, City University of New York, 149th St, Flushing, NY 11367, USA
This is the peer reviewed version of the following article: Alyokhin, A.V., B.M. Rosenthal, D.C. Weber, and M.B. Baker. 2025. Towards a unified approach in managing resistance to vaccines, drugs, and pesticides. Biological Reviews 100: 1067-1082, which has been published in final form at https://doi.org/10.1111/brv.13174. This article may be used for non-commercial purposes in accordance with Wiley Terms and Conditions for Use of Self-Archived Versions. This article may not be enhanced, enriched or otherwise transformed into a derivative work, without express permission from Wiley or by statutory rights under applicable legislation. Copyright notices must not be removed, obscured or modified. The article must be linked to Wiley’s version of record on Wiley Online Library and any embedding, framing or otherwise making available the article or pages thereof by third parties from platforms, services and websites other than Wiley Online Library must be prohibited.
ABSTRACT
Everywhere, pests and pathogens evolve resistance to our control efforts, impairing human health and welfare. Developing sustainable solutions to this problem requires working with evolved immune and ecological systems, rather than against these evolutionary forces. We advocate a transdisciplinary approach to resistance based on an evolutionary foundation informed by the concepts of integrated pest management and One Health. Diverse, multimodal management approaches create a more challenging environment for the evolution of resistance. Given our permanent evolutionary and ecological relationships with pests and pathogens, responses to most biological threats to health and agriculture should seek sustainable harm reduction rather than eradication.
Key words: resistance, evolution, integrated pest management, One Health, vaccines, pesticides.
I. INTRODUCTION
Pests and pathogens represent dominant cultural and economic concerns, as vividly evidenced by the recent COVID-19 pandemic. They drive our biological evolution and technological and social development, as do our efforts to manage them. Our dynamic interactions with pests and pathogens entail reciprocal adaptations sometimes conceived as an evolutionary arms race. At the same time, seeking ‘magic bullets’ to eradicate pests removes pest control from its proper ecological and evolutionary context. Separating ourselves from the rest of the biosphere in this way has a rich, and sometimes tragic, history of failure.
We generally define pests as organisms that interfere with something that we value (Kogan & Jepson, 2007). This anthropocentric definition disregards our (often negative) impacts on other species (Geier, Clark & Briese, 1983) but is well entrenched in present-day discourse. Our pests can cause us direct harm by extracting resources by consuming parts of our bodies. They can also degrade systems upon which we rely, and compete with us for resources (Kogan & Jepson, 2007).
It is common to group microscopic parasitic pests separately under the name of pathogens. However, there is no compelling biological reason for doing so. Pathogens, although smaller, cause physiological and evolutionary harm to human health and welfare that is similar to that caused by larger organisms. Acknowledging this unity is important for building transdisciplinary connections between agriculture, natural resource management, and urban management on the one hand, and epidemiology, public health, and medicine on the other. We use both terms throughout this review to honour current conventions prevalent in different fields of science, but with an understanding that there is no distinct dichotomy between them. Here, we explore patterns of success and failure in managing pests and pathogens, harnessing insights from diverse biological disciplines.
Sustainably managing pests and pathogens requires diverse and flexible systems, modelled on natural evolutionary processes, that have a built-in capability of responding to ever-changing pressures. Vaccination offers such a harm-reduction paradigm. Expanding this paradigm from an organismal to community and ecosystem levels may be useful in structuring sustainable efforts to manage agricultural and other landscapes beset by loss to pests (including weeds) and pathogens. The main goal of this review is to discuss the potential for such a shift in the face of its numerous challenges.
II. DEFINITIONS OF RESISTANCE
Broad availability of effective pest and pathogen controls has decreased human mortality and improved the average quality of life (Cooper & Dobson, 2007; Flasche & Atkins, 2018). However, the sustainability of these unquestionably positive developments is at risk because the evolution of resistance threatens this success (Gould, Brown & Kuzma, 2018; Alyokhin et al., 2022; Adebisi, 2023; Mortensen, Ryan & Smith, 2024). We are approaching an end to the era of efficient and readily available chemical solutions to problems created by pests and pathogens (Alyokhin et al., 2015; Bäumler, 2024). Non-target effects of control measures that degrade the environment also deserve attention but lie outside our present scope.
Resistance has been defined either as a statistically detectable increase in frequency of resistant alleles in affected populations, or as a control technique’s failure to suppress target pest or pathogen populations to acceptable levels (Tabashnik et al., 2014). All fields concerned with resistance evolution share a tension between these two definitions. Earlier definitions of resistance were explicitly based on the ability to detect a shift in survival of a pest in laboratory assays (National Research Council, 1986). However, a control agent may continue to provide real-world protective value after detection of resistance via bioassay. Thus, resistance may practically be understood as a level of field-evolved resistance that impacts control (Tabashnik et al., 2014), leading to increased costs of protection, lower yields, or abandonment of the crop or protection method. Field-evolved resistance contrasts with laboratory-selected resistance evolved under artificial conditions (Tabashnik et al., 2014). The latter illustrates the possibility and possible mechanisms of a tool’s loss of practical utility but does not necessarily mimic field developments. Resistance to vaccines may also be recognized either as increased ability to replicate in a vaccinated host (Kennedy & Read, 2017), as evolved loss of the ability to provide clinical benefit (Clemens et al., 1996; Shim & Galvani, 2012), or as loss of the vaccine’s ability to diminish an infected person’s ability to transmit the infection to others. Such ways of conceiving resistance are not mutually exclusive; resistance entails a genetic process with multiple practical implications.
III. PEST AND PATHOGEN MANAGEMENT
With the possible exception of Archaea (Eckburg, Lepp & Relman, 2003; Bang & Schmitz, 2015), pests range widely over all major taxa of living organisms. (As discussed above, microscopic parasites are often referred to as pathogens). This diversity results in a wide range of taxon-specific traits that need to be considered when devising management techniques. Unfortunately, it also thwarts the development of a ‘one size fits all’ approach. Still, there are certain commonalities in dealing with undesirable species across the entire spectrum of life.
A wide variety of tactics seek to kill, prevent, suppress, or otherwise manage pests (Mason, 2021): (1) chemical control employs poisons (usually referred to as pesticides, drugs or antibiotics) to kill pest organisms. (2) Biological control employs living organisms to manage pest populations, harnessing predation, parasitism, and competition to advance conservation and production goals by augmenting, or introducing, natural enemies. Although often not considered to be biological control, phage therapy, probiotics, and faecal transplantation also fall under this category. (3) Environmental manipulation encompasses cultural control in crops (crop rotation, timing, tillage, irrigation, hand weeding, sorting and winnowing, etc.), mechanical exclusion (crop row covers, window screens, hermetic storage of grains, water filtration, human face masks), heat treatments, and irradiation. (4) Vaccination, selective breeding, and genetic engineering enhance host resistance and impede the transmission of pathogens. (5) Quarantines prevent colonization of new hosts and habitats by pests. (6) Behavioural manipulation of pest organisms uses semiochemicals (pheromones, kairomones, allomones) as attractants, repellents, or confusants to move, entrap, or disrupt reproduction of animal pests. (7) Other forms of pest mating disruption involve releases of sterilized or genetically altered male insects to suppress natural pest reproduction, as the sterile-insect technique and gene-drive tactics, respectively.
Pest and pathogen management tactics can follow two broad strategies: proactive (or preventative) versus reactive (or therapeutic) (Vandermeer & Andow, 1986). The former aims to prevent a problem from occurring, while the latter aims to deal with a problem once it happens. Conventional wisdom holds that ‘an ounce of prevention is worth a pound of cure’. However, this oversimplification does not hold true in all situations. Unnecessary prophylactic treatments with significant side effects can cause needless harm. On the contrary, reactive treatments, triggered by rational thresholds for action, can limit intervention to cases where it is needed. Thereby, they may reduce expenses and collateral damage to the environment (Vandermeer & Andow, 1986; Higley & Pedigo, 1993).
Another choice facing stakeholders pits eradication against maintenance of pest populations below damaging levels (Suckling et al., 2014). When successful, eradication brings high reward. Unfortunately, most pest species are characterized by high fecundity and short generation times. Therefore, high growth rates of their populations enable a few survivors to seed rapid resurgence. Overestimating eradication’s achievability risks wasting resources and incurring extensive environmental damage (Booy et al., 2020; Green & Grosholz, 2021). Unsuccessful eradication attempts may also promote resistance by applying high selection pressure to target populations. Decisionmakers should honestly assess the likeliest outcome, but doing so can reach unpopular conclusions. Aggravating this, such assessments are often fraught with high uncertainty.
Eradication may work well on a small and medium scale, especially for non-native species with limited distributions in newly invaded areas (Simberloff, 2001). Few large-scale eradication efforts succeed. Smallpox represents an instructive exception. It is a deadly disease of humans caused by Variola virus with mortality rates exceeding 20%. Smallpox was eradicated via a large international program carried out under the auspices of the World Health Organization in 1959–1977. The program was based on a combination of extensive vaccination of at-risk populations and quarantine measures in affected areas. Its success was facilitated by the lack of animal reservoirs, the infrequency of asymptomatic infections, and the effectiveness of vaccination coupled with quarantine (Meyer, Ehmann & Smith, 2020). Another exceptional control program eliminated invasive cotton boll weevil, Anthonomus grandis, from the USA. In that case, targeted insecticide applications were combined with captive-reared sterile male release, distribution of poison baits, and careful monitoring of weevil populations with pheromone traps (Allen, 2008; Raszick, 2021).
Pest and pathogen management ranges from more-or-less indiscriminate treatment of current or potential infestation sites to targeted delivery to afflicted, or imminently at-risk, individuals. Either approach can occur at a variety of spatial scales (Faust, 2008). Because of possible damage to non-target organisms, the widespread residual effects of control techniques that are used on a broad scale are undesirable. These result not only from direct application, such as pesticide spraying, but also from excretion of incompletely metabolized drugs (Shalini et al., 2010; Quesada et al., 2019; Ojemaye & Petrik, 2019). We know of no such ancillary harms from vaccines (He et al., 2016).
Resistance to antibiotics and pesticides constitutes a major constraint on human well-being. Drug-resistant bacterial infections were associated with an estimated 4.95 million deaths in 2019, directly causing an estimated 1.27 million of those (Murray et al., 2022). Surveys document 523 cases of herbicide resistance in 99 crops in 72 countries for 269 species of weeds. These involve 167 different herbicides possessing 21 known modes of action (Heap, 2023). Resistance to insecticides and acaricides has been reported for 391 different chemicals in 23 orders of arthropods (Mota-Sanchez & Wise, 2023). The recurrent failure of pesticides and antibiotics is an evolutionary consequence that logically follows from their widespread use (Alyokhin et al., 2022).
Control strategies other than chemicals also induce evolutionary responses because adaptation to unfavourable environments constitutes a fundamental property of life (Tu & Rappel, 2018; Onstad & Knolhoff, 2023). For example, hand pulling barnyard grass, Echinochloa crus-galli, selected for morphological and phenological traits rendering the weed difficult to distinguish visually from rice, Oryza sativa (Ye et al., 2019). Visual removal of potato, Solanum tuberosum, plants demarcated by the O strain of Potato virus Y selected for viral strains (NTN and Wilga) producing no such visible damage (Gray et al., 2010). Introducing myxoma virus to control invasive European rabbits, Oryctolagus cuniculus, in Australia selected for resistance to this virus (Alves et al., 2019). Widely applying granulosis virus to European apple orchards induced resistance in the codling moth, Cydia pomonella (Asser-Kaiser et al., 2007). Green peach aphid, Myzus persicae, evolved resistance to the parasitoid wasp, Aphidius colemani, commonly released for its control in commercial greenhouses. Resistant aphids suffered fewer wasp stings, and a lower percentage of those stings resulted in successful parasitoid development (Herzog, Müller & Vorburger, 2007; Gillespie et al., 2009). Northern corn rootworm, Diabrotica barberi, eventually overcame annual crop rotations between corn and soybean by extending its diapause for an extra year, enabling it to thrive where corn was planted only every other year (Levine, Oloumi-Sadeghi & Fisher, 1992). Western corn rootworm, Diabrotica virgifera virgifera, switched its egg laying to soybean fields, enabling hatching larvae to exploit the corn planted the following year (Levine et al., 2002).
There are also several recorded cases of vaccine failures due to resistance of the target pathogen, including influenza, pertussis, pneumococci, and hepatitis B. However, compared to literally hundreds of cases reported for failing pesticides and antibiotics, such cases appear to be relatively rare. Furthermore, resistance to vaccines typically takes longer to develop, especially in comparison to antimicrobial drug resistance (Kennedy & Read, 2017, 2018). A notable, and unfortunate, exception is the rapidly mutating influenza virus that evolves quickly enough to necessitate annual reformulation of the vaccine (Sah et al., 2018). This natural antigenic evolution may be further exacerbated by viral adaptation to being grown within chicken eggs as a part of the mass manufacturing process of the inactivated influenza vaccines (Zost et al., 2017).
IV. SUSTAINABILITY, STABILITY, AND HOMEOSTASIS
To be sustainable, the development of a human civilization must meet the needs of the present generations without compromising the ability of future generations to meet their own needs (The Brundtland Commission, 1987). Sustainable systems must be capable of withstanding an array of disturbances, in particular those threatening their productivity (Conway, 1993), thus remaining stable over an extended period of time. Although sustainability is usually discussed within the framework of human activity, the level of stability is also an important parameter defining the state of natural systems.
At an ecosystem level, stability refers to the ability of a community of living organisms to retain its structure and function within a narrow range surrounding a constant baseline in the face of natural or human-related environmental disturbances (Van Meerbeek, Jucker & Svenning, 2021; Segrestin et al., 2024). At an organismal level, stability is usually referred to as homeostasis. It is defined as a self-regulating process by which an organism can maintain its internal conditions around a more-or-less constant baseline while adjusting to changing and often unfavourable external conditions (Billman, 2020). Stability does not necessarily equal a lack of change: it may result from a relatively quick return to the original range of parameters following a perturbation (Stiling, 2002).
Stability depends on intricate interactions among diverse components guided by feedback loops. It is often compromised in anthropogenic systems because they are usually characterized by high levels of disturbance. This commonly results in a pest outbreak or an onset of disease, which constitutes a sustained deviation from the constant long-term baseline. Therefore, human intervention to prevent or suppress these undesirable developments can be viewed as an effort to maintain system stability. Pest management systems that are diverse both in terms of control methods and environmental conditions have a better chance of being generally more stable, just as natural ecosystems with high biodiversity are often more stable than ecosystems with low biodiversity (Loreau & de Mazancourt, 2013).
V. SUSCEPTIBILITY, A NATURAL RESOURCE
The susceptibility of pests and pathogens to control measures constitutes a valuable natural resource, fulfilling human needs (Hueth & Regev, 1974). While resistance alleles remain rare, control measures work (Tabashnik, 1994; Andow & Alstad, 1998). Unfortunately, widespread control efforts deplete susceptibility (Davis & Frisvold, 2017; Doole & James, 2023).
Susceptibility is typically, but not always, a non-renewable resource (Hueth & Regev, 1974; Mitchell & Onstad, 2023; Doole & James, 2023). Only rarely do resistant populations revert to susceptibility once selection pressure is removed (Abedi & Brown, 1960; Tabashnik, 1994; Meka et al., 2004; Sullivan et al., 2013; Leathwick, Ganesh & Waghorn, 2015; Ocan et al., 2019). Such reversion requires that resistance genes reduce relative fitness in the absence of pressure (Tabashnik, 1994; Sullivan et al., 2013; Délye, Jasieniuk & Corre, 2013; Gould et al., 2018). Moreover, it may prove incomplete (Tabashnik, 1994; Susser et al., 2011; Alyokhin et al., 2015), and revertant populations rapidly evolve resistance when subjected to pressure anew (Tabashnik, 1994; Nascimento, Lira & Torres, 2023).
VI. EVOLUTIONARY AND GENETIC MECHANISMS OF RESISTANCE
Resistance to control measures exemplifies directional selection disfavouring susceptible genotypes and favouring resistant genotypes (Alyokhin et al., 2022). Control efforts may also inadvertently kill a pest’s natural enemies, further favouring resistant survivors (Bosch, 1978). This shifts the mean value of the resistance trait. In some instances, selection by one agent may produce resistance to other agents (a phenomenon known as cross-resistance). The risk of cross-resistance increases for chemicals with similar molecular structures and/or modes of action (Alyokhin et al., 2022). However, the same adaptation can also effect resistance to rather different stressors, such as heat shock and exposure to xenobiotics (Gould et al., 2018; Pu, Wang & Chung, 2020).
Genetic bases of resistance can vary along axes of dominance, number of loci, coding versus regulatory changes, and mode of action. Dominance in diploid eukaryotes strongly influences how stressors select for rare resistance alleles (Gould, 1998), and aids crop refuges as a means to slow resistance evolution (Tabashnik, Brevault & Carriere, 2013; Tabashnik, Fabrick & Carrière, 2023). Recessive inheritance greatly slows the rate of increase of very rare novel mutations, but has less effect on more common alleles, such as those maintained for other functions.
High initial frequency, even in haploid taxa, should greatly accelerate resistance spread. For example, many antibiotic resistance genes long pre-date human pharmacological use (Spagnolo, Trujillo & Dennehy, 2021). Bacteria produce detoxifying enzymes in response to naturally occurring toxins produced by other microorganisms present in the same environments. They can also share these genes widely (Peterson & Kaur, 2018). These enzymes serve as a foundation for resistance to human-made antibiotics.
High-level resistance to xenobiotics may result from single mutations, of large effect, inducing changes at a target site of the xenobiotic (Feyereisen, Dermauw & Van Leeuwen, 2015). High-level, field-evolved resistance often results from one or a few such mutations (Roush & McKenzie, 1987; Oakeshott et al., 2013), which do not incur the energetic cost of producing more enzymes. However, the importance of other types of genetic change, including polygenic resistance, up- and down-regulation of gene expression, or duplication of existing genes, should not be underestimated (Feyereisen et al., 2015). In particular, transcriptional studies often reveal many contributions to resistance phenotypes (Clements et al., 2016). Gene amplification and duplication commonly drive resistance evolution in insects (Bass & Field, 2011). Duplicate copies also allow haploid taxa to produce new resistance phenotypes and allow diploid taxa to fix phenotypes originally expressed as heterozygous genotypes (Labbé et al., 2014). Resulting physiological mechanisms of resistance include detoxification (Nauen et al., 2022), transport (Dermauw & Van Leeuwen, 2014), cuticular thickening (Dang et al., 2017), sequestration, and excretion (Panini et al., 2016). Resistance may also arise from heritable changes in behaviours, such as movement, feeding, entering and terminating diapause, oviposition, etc. (Onstad & Knolhoff, 2023). An immensely consequential work on the factors limiting corn and soybean production in the midwestern USA showed that the evolution of a novel detoxifying pathway (rather than targeted mutations for specific pressures) explains the spread of metabolic resistance in a common weed to a range of pesticides (Tranel, 2021).
Fewer mechanisms of release from vaccines have been identified, but that may be a result of fewer examples to study. Pathogens may evade host immune responses through modifications in their antigens, may produce substances that suppress host immunity, and/or may overwhelm host immunity by enhancing reproduction (Day et al., 2022). For example, selection against a generalized immune simulant, flagellin in Yersinia ruckeri, may have driven a decline in motile forms of that parasite, although the vaccine seemingly targets the cell wall, not the flagellum (Welch & LaPatra, 2016; Kennedy & Read, 2018). In other cases, one or many changes in the region targeted by the vaccine overcomes protection from infection or severe illness (Deb, Vilvadrinath & Goel, 2021). In theory, any means to counter natural immunity may conceivably also counter vaccine-induced immunity.
Selection of resistance genes from the standing genetic variation or from new mutations is the most generally acknowledged mechanism responsible for the evolution of resistance in populations of pests and pathogens. However, that does not entirely explain the advent of resistance. Some environmentally induced heritable changes in gene expression do not derive solely from changes in DNA sequences. Abedi & Brown (1960) questioned whether a single generation of selection could produce dichlorodiphenyltrichloroethane (DDT) resistance in the yellow fever mosquito, Aedes aegypti, suspecting that toxin exposure induced expression of pre-existing physiological responses. Later experiments with the congener mosquito Ae. albopictus showed that changes in DNA methylation were indeed linked to reduced susceptibility to the insecticide imidacloprid (Oppold et al., 2015). Similarly, Escherichia coli exposed to the antibiotics ampicillin, tetracycline, and nalidixic acid developed resistance and then reversed to susceptibility quicker than could have been plausibly expected based on random mutation and selection alone (Adam et al., 2008). Brevik et al. (2021) further conceptualized that successive waves of selection should reduce the size and genetic variability of pest populations, eroding their potential to evolve resistance to subsequent insecticides. In practice, we see no such erosion (Alyokhin et al., 2015).
Epigenetic modifications, including DNA methylation, RNA-associated silencing, and histone modification (Guerrero-Bosagna & Skinner, 2012), likely promote resistance acquisition in populations of pests and pathogens. In the green peach aphid, methylation of the esterase genes that detoxify insecticides increased their expression and decreased aphid susceptibility to insecticides (Field et al., 1989). In the Colorado potato beetle Leptinotarsa decemlineata insecticide exposure also changed methylation of resistance genes, such as cytochrome P450s (Brevik et al., 2021). In both cases, pests retained methylation patterns over several generations (Field et al., 1989; Brevik et al., 2021). Histone modification in the malaria parasite Plasmodium falciparum reduces uptake of antimalarial drugs (Sharma et al., 2013; Mira-Martínez et al., 2019). Histone acetylation influences drug resistance in the opportunistic pathogen Candida albicans; RNA silencing influences such phenotypes in the fungal human pathogen Mucor circinelloides and in the model fission yeast, Schizosaccharomyces pombe (Chang et al., 2019). Methylation also plays a role in antibiotic resistance of Neisseria meningitidis, a bacterial cause of human meningitis (Ghosh et al., 2020). Methylation patterns differ in populations of horseweed, Conyza canadensis, resistant or susceptible to glyphosate (Margaritopoulou et al., 2018). Moreover, antibiotic administration has been shown to induce broad changes in gene expression, including virulence factors and systems that can increase exogenous DNA uptake (Holman et al., 2018).
Epigenetics is a relatively new field. Therefore, its importance for the development of pesticide and drug resistance is not fully understood, especially in comparison to Darwinian selection. However, there is mounting evidence that epigenetic mechanisms are responsible for the evolution of practically significant resistance at least in some cases (Oppold & Müller, 2017; Markus et al., 2018; Ghosh et al., 2020; Patra et al., 2022; Mogilicherla & Roy, 2023). Currently, epigenetic resistance is not included in most mathematical or conceptual models of resistance, which assume resistance arises via multi-generational selection (Li et al., 2021; Onstad, 2023). This is unfortunate, because epigenetic processes do not induce genetic bottlenecks, and thus permit vastly accelerated phenotypic responses. These attributes may undermine pest management approaches (such as combining high selection pressure with untreated refuges). Fully incorporating epigenetic mechanisms into a bigger picture of pest resistance would afford a better understanding of the dynamics of resistance and might suggest better management approaches.
VII. COEVOLUTION
Interspecific interactions drive biological evolution. Coevolution results when traits in one population change in response to traits in other populations (Ehrlich & Raven, 1964). For example, herbivorous insects evolve enzymes enabling them to consume plant tissues laden with a toxin. In turn, the toxin may change in ways that resist the action of the detoxifying enzyme, restoring plant toxicity. Coevolution has been often proposed as the major factor responsible for the biological diversity on our planet (Laine, 2009). However, no pairwise interaction exists in isolation. Therefore, diversification of biological traits may be better considered a multilateral process (Suchan & Alvarez, 2015).
Because such interactions have always driven biological evolution, human technological advances act on pest populations that have amassed defences against diverse threats. Systems underlying insect resistance to plant toxins underly insecticide resistance, for example (Alyokhin & Chen, 2017). Similarly, most antibiotics in clinical use derive from natural microbial products. Resistance to these compounds also evolved in natural ecosystems long ago, with many clinically problematic resistance genes being similar to those found in nature (Chait, Vetsigian & Kishony, 2012).
Modern technologies diminish the selective advantage for human genotypes conferring resistance to pests and pathogens. We now primarily rely on broadly efficacious hygiene measures, vaccines, and specific synthetic chemicals to mitigate harms. Yet, these recent developments do not alter the evolutionary nature of the interaction. Pest and pathogen genotypes exhibiting resistance to new drugs can outcompete susceptible strains, leading to control failure. The failure of first-line chemicals can then lead to the adoption of less efficacious, more toxic, and more expensive chemicals. Moreover, pathogens resistant to certain drugs are predisposed to resist entire classes of drugs. The advent of multi-drug resistant tuberculosis especially exemplifies this problem (Singh & Chibale, 2021). The ensuing series of reciprocal changes parallels coevolutionary processes found in nature (Goeschl & Swanson, 2002; Alyokhin et al., 2015). Yet we deploy our agents of selection with a high degree of hubris and an apparently diminishing capability to adapt to the pest’s resistant reaction.
VIII. VACCINATION – A MODEL TO FOLLOW
Vaccines boast a more sustainable track record than pesticides or antibiotics. Vaccine use and natural immunity do favour survival of “immune escape’ variants (Carabelli et al., 2023), undermining long-term efficacy against given genotypes or serotypes of that pathogen. However, resistance appears rare despite widespread use (Kennedy & Read, 2017, 2018). This rarity likely derives from five interacting characteristics inherent to how vaccines work.
Firstly, exquisitely specific responses induced by vaccines help individuals and populations mitigate harms wrought by very specific infections (Davies & Davies, 2010). Although some vaccines may confer benefits beyond their intended use (Aaby, Netea & Benn, 2023), we are unaware of vaccines rendering non-target pathogen species recalcitrant to management. By contrast, even narrow-spectrum antibiotics and pesticides affect, and hasten resistance in, unintended targets (Tedijanto et al., 2018; Gage & Schwartz-Lazaro, 2019; Singh & Leppanen, 2020; Pisa et al., 2021; Fishbein, Mahmud & Dantas, 2023). Vaccine administration does not appear to favour cross-species horizontal transmission of resistance, which foreshortens the useful life of antibiotics and pesticides (Davies & Davies, 2010; Beckie et al., 2021; Dimitriu, Matthews & Buckling, 2021). Although vaccine specificity slows progress towards new vaccines (because each pathogen requires tailored solutions; Kim, Choi & Shin, 2023), new platforms speed up the development and deployment of new vaccines (Hogan & Pardi, 2022; Coughlan, Kremer & Shayakhmetov, 2022), at least partially ameliorating this problem.
Secondly, ‘immunological memory’ defines a body’s (and therefore, population’s) response to vaccine administration (Sharrock & Sun, 2020; Lipsitch et al., 2020). Effective vaccines prime memory T-cells and memory B-cells, hastening responses to subsequent exposures. Such memory generally benefits the recipient, although ‘original antigenic sin’ may impair responses to changing threats (Shukla et al., 2020; Yewdell & Santos, 2021). Vaccine boosters further increase antibody titres; when reformulated, they may also better target recently evolved variants (Burckhardt et al., 2022). By contrast, chemical pesticides do not ‘remember’ prior bouts with pests and cannot remain ready to protect a field from future incursions. In this way, they differ from the memory B and T cells that persist in a body and in a population. Unfortunately, pest populations do ‘remember’ prior applications of pesticides (in the form of resistance alleles that modify their physiology or behaviour). These alleles seldom subside to pre-exposure frequencies, even after cessation of the pesticide’s use, despite fitness costs that resistance alleles can impose in the absence of pesticide pressure (MacLean et al., 2010; Freeman et al., 2021). Cycled antibiotics typically suffer this same fate; once established, antibiotic resistance seldom if ever truly goes away (Salyers & Amábile-Cuevas, 1997).
Third, vaccines activate a complex and diverse suite of defence systems, whereas antibiotics and pesticides typically interfere with rather specific physiological processes. This difference shapes the nature of possible evolutionary responses. Responses to vaccines of varying complexity differ (Zimmermann & Curtis, 2019). Simple recombinant vaccines may express just one or a few epitopes (protein motifs bound by particular antibodies); more complex vaccines may entail complex mixtures, dead formulations of entire pathogens, or even live attenuated pathogen strains. Regardless, hosts respond to vaccines by triggering both: (1) innate immune responses (including inflammatory responses and the complement system, and cytokine signalling that recruits ‘generalist’ macrophages that ingest pathogens and hasten presentation of pathogen antigens to other immune system components); and (2) adaptive immune responses (honing and priming populations of T and B cells that specifically recognize and strongly bind particular pathogens (Roth et al., 2022).
Complex responses pose a fundamental evolutionary challenge to pathogens. This is generally not the case for pests exposed to, for example, a single chemical toxin. Detoxifying enzymes (Lu, Song & Zeng, 2021) and efflux pumps (Henderson et al., 2021) can mitigate diverse chemical threats. On the contrary, the varied components of immunity present an array of assaults for which no single solution (except for evading detection) likely exists. By preventing infections, vaccines can reduce reliance on antibiotics, thereby helping forestall and manage resistance (Micoli et al., 2021).
Pathogens (especially those with rapid evolutionary rates and recombining antigens, like influenza) certainly do generate variants capable of rendering this year’s vaccines less efficacious next year (Wei et al., 2020). Also, certain pathogen strains (and cancerous tumours; Jhunjhunwala, Hammer & Delamarre, 2021) do evade detection (Carlson et al., 2015). For example, immune evasion may follow the depletion of T cells in people with acquired immune deficiency syndrome (AIDS) (Nowak, May & Anderson, 1990). However, evading detection in one person does not necessarily help a pathogen evade detection in the next person. Individuals harbour distinct molecules (human leukocyte antigens) that bind and present antigens to B and T cells (Muraduzzaman et al., 2022). People generally do not share the same ‘blind spots’ because diversifying selection maintains huge stores of allelic variation in the molecules that bind and present antigens. This lessens one’s resemblance (in this respect) to one’s neighbours or even one’s kin (Radwan et al., 2020). Consequently, vaccine recipients recruit distinct effector molecules and cells, precluding or slowing ‘vaccine escape’ for entire populations.
By contrast, one weed’s successful response to an herbicide likely does not depend on such contextual nuances; indeed, resistance developed in response to one herbicide can predispose a plant to resistance to other herbicides (Bobadilla & Tranel, 2024). The traits conferring resistance in one field will confer resistance in neighbouring fields. Expression of resistance may transcend cultivars and even cross species boundaries. Industrial monoculture exacerbates this likelihood. These challenges test our cultural, agronomic, and technological creativity (Perotti et al., 2020).
Fourth, vaccines induce expansion and refinement of immune cells that meet the demands of a dynamic challenge. From a vast repository of B and T cells, vaccines (and pathogens) induce competition that favours expansion of those that bind best, and favour [through variable diversity joining (VDJ) recombination; Chi, Li & Qiu, 2020] elaboration of new B and T cell receptors that bind such targets even better (Joglekar & Li, 2021; Laidlaw & Ellebedy, 2022). Expanding and honing these cells poises the vaccine recipient to recognize and respond to infection. By contrast, pharmaceutical companies test vast libraries of molecules for antibiotic activity (Krell & Matilla, 2022); but once deployed, non-living antibiotics cannot adapt to changing demands; nor do we spray variable pesticides capable of competing for primacy and adjusting to changing conditions.
The dynamism characteristic of vaccine-induced immunity does not define application of static management techniques. However, it may characterize attempts to control pests using integrated pest management, living biological control agents, and bacteriophage therapy as discussed below. For example, practitioners of Darwinian beekeeping treat honeybee colonies as suites of adaptations shaped by natural selection and maintain colony health by allowing the bees to function as naturally as possible (Seeley, 2017).
Finally, prophylactic vaccine administration may diminish the size of pathogen populations and induce population bottlenecks that lower their genetic diversity, thereby slowing evolutionary responses (Kennedy & Read, 2017, 2018; Goldman, 2021). Applications of pesticides and drugs will likely produce a similar result. Unfortunately, most of them also have large non-target effects. Such effects risk human injury and environmental damage. Furthermore, they weaken the ‘immunity’ of ecosystems by killing existing natural enemies. As a result, prophylactic application of most pesticides and antibiotics is not advisable.
Vaccination provides insights relevant for sustainable harm reduction, reinforcing the experience of those promoting ecological and evolutionary principles to guide efforts countering losses from pests. The multiplicity of effector systems inherent to any immune response resembles the ‘multiple small hammers’ advocated by those seeking to prevent harms wrought by weeds through varied tactics applied, throughout the year, to limit the production, dissemination, and germination of weed seeds (Bagavathiannan & Davis, 2018). Complex ecological systems, like complex immune systems, entail redundancies that defy rapid circumvention. Mimicking immunity, by incorporating multiple natural adaptive strategies, enables pest and pathogen management systems to address today’s problem while anticipating tomorrow’s.
IX. RESISTANCE MANAGEMENT
Currently, the most common approach to resistance management diversifies selection pressures on pest and pathogen populations by alternating application of active ingredients or by simultaneous use of tank mixes, pyramided toxins in transgenic plants, and drug cocktails (Raymond, 2019). Either approach can be performed proactively, while target populations are still susceptible to each active ingredient, or reactively, after a particular chemical active ingredient begins to fail and must be replaced or supplemented by other active ingredients. Despite long-established advantages of mixing toxins to prevent or delay resistance (Tabashnik et al., 2023) and despite a tremendous diversity of metabolic pathways amenable to transgenic modification (Gatehouse, 2013), conventional agriculture does not yet widely implement multimodal, flexible, and preventative control measures. The number of genetically modified species and the mechanisms of their defences against pests remain very low. Mixing ingredients after the advent of resistance instead reacts to a foreseeable problem. Furthermore, pyramiding constitutively expressed toxins risks excessive exposure.
Another approach is to relax selection pressure on target populations by leaving untreated refuges that allow susceptible phenotypes to survive, thus decreasing the overall frequency of resistant alleles in a population (Onstad & Knolhoff, 2023). Refuges are mandated for transgenic plants that express toxins protecting them from insect feeding. They can be structured when a certain percentage of crop area is deliberately set aside as an untreated refuge, or unstructured (natural) when an otherwise unmanaged habitat serves as a source of susceptible individuals (Huang, Andow & Buschman, 2011; Knight, Head & Rogers, 2021).
Using refuges is uncommon except with transgenic crops because of understandable reluctance deliberately to encourage certain levels of pest damage, even though there are examples of crop refuges with lower overall damage levels than uniform treatment (Zhu et al., 2000). Also, the ethics of leaving part of a population unprotected may be questionable (Adebisi, 2023). This is not very important when dealing with plants unless there is a need to prevent imminent famine. However, it is certainly a consideration for humans and, potentially, for other animals.
The opposite of a refuge is a reservoir, which is when the use of a treatment outside one target protected population selects for resistance that rebounds to the target interaction. The use of antibiotics for growth promotion in domestic animals is one clear example (Unno et al., 2010; Looft et al., 2012; Caniça et al., 2015).
Most vaccine programs seek to limit harms and enhance health in the face of continued pathogen transmission, rather than seeking disease eradication. Unvaccinated individuals lessen the strength of selection for resistance (or evasion) to such vaccines, but may support a large and diverse pool of pathogens, thereby increasing the pool of resistant mutations (Kennedy & Read, 2017, 2018; Goldman, 2021). Unlike typical crop refuges, unvaccinated individuals retain immune responses that shape pathogen evolution, including favouring phenotypes that evade recognition. The diversity and dynamism of immune responses also make them less prone to failure compared to pesticides and drugs, even in the presence of a strong selection pressure (Kennedy & Read, 2017, 2018).
Integrated pest management (IPM) is a knowledge-based, systematic, and coordinated attempt to employ multiple control techniques to reduce pest damage to economically acceptable levels while minimizing negative environmental impacts (Prokopy, 1993; Zalom, 1993). Using IPM is the most effective way to delay resistance because it combines different control techniques, reduces selection pressure, and eliminates unnecessary treatments (Onstad & Knolhoff, 2023).
Integrated weed management, as an IPM example, seeks to sustain success via ecological principles that refocus efforts from immediate yields to long-term and regional prosperity, anticipating evolutionary responses and focusing attention on the size and composition of the weed seed bank (Bagavathiannan & Davis, 2018). Adopting multiple management approaches, deployed throughout the life cycle, and making use of natural barriers to seed production, dispersal, and germination, offers advantages over widespread and repeated use of any one technology. A large empirical test found that more diversified crop rotation systems can maintain yields while decreasing dependency on external synthetic fertilizers, herbicides, and fossil fuels (Davis et al., 2012).
Genetic changes conferring simultaneous resistance to vastly different control techniques used under IPM are unlikely (Alyokhin et al., 2015, 2022). However, they are not entirely impossible. Widespread use of insecticide-treated mosquito bed nets induced nocturnal mosquito vectors of human malaria in Africa, Anopheles gambiae and A. funestus, to switch to feeding outdoors during the daytime (Sherrard-Smith et al., 2019). This change constituted simultaneous behavioural resistance both to physical control (window screens and bed nets) and chemical control (insecticides used to treat bed nets). This IPM system was rather simple, but still fits the definition (Prokopy, 1993). Therefore, the reported adaptation should serve as another reality check: we must not become overconfident about our ability to control nature. Integration more advanced than simple combinations of two different techniques may be needed to prevent failures due to the evolution of resistance.
IPM philosophically parallels the concept of One Health, which recognizes that the health of humans, animals, plants, and ecosystems are interconnected. As a result, One Health uses coordinated, collaborative, multidisciplinary and cross-sectoral approaches to address potential or existing health risks originating at the animal–human–ecosystems interface (Mackenzie & Jeggo, 2019). This approach manages antibiotic resistance through integrated interventions that include alternative drug treatments, vaccinations, better sanitation to prevent transmission, reduction in the amount of drug residues entering the environment, and biological control using susceptible competitors and/or bacteriophages (Hernando-Amado et al., 2019).
Because long-term susceptibility of pest and pathogen populations is a public good, its preservation must be addressed from a societal perspective within the appropriate regulatory frameworks (Hu, 2020; Haywood et al., 2021; Shaw et al., 2023). For instance, the British Society for Antimicrobial Chemotherapy, the Hospital Infection Society, and the Infection Control Nurses Association, on request from the Department of Health’s Specialist Advisory Committee on Antimicrobial Resistance, developed guidelines on how to deal with methicillin-resistant Staphylococcus aureus (MRSA). The guidelines integrated comprehensive monitoring for MRSA occurrence, decreased selection pressure through judicious use of antibiotics, quarantine, sanitation, and adequate staffing by nursing personnel (Coia et al., 2006). They were codified in the Health Act of 2006 and eventually led to a 2.27-fold decrease in MRSA infections (Robotham et al., 2016). In another example, leaving non-transgenic refuges is required for insect-protected crops that express Bacillus thuringiensis (Bt) toxins, both by license agreements with seed producers and by government regulations. Analysis of an extensive data set generated by 77 studies conducted on five continents (Tabashnik et al., 2013) confirmed that, when properly enforced, this requirement indeed maintains susceptibility of the targeted pest populations.
In some cases, grassroots-level collaborations on local and regional levels may be more effective than overarching government mandates (Gould et al., 2018; Shaw et al., 2023). The Arizona Cotton Research and Protection Council, a commercial grower organization, developed, funded, and implemented an IPM plan for the pink bollworm, Pectinophora gossypiella, in collaboration with the US Department of Agriculture and the US Environmental Protection Agency. As a result, bollworm populations collapsed by more than 99% and no resistance was detected (Tabashnik et al., 2010). On an even smaller scale, collaboration among farmers in western Maine, USA, Bayer CropScience, and the University of Maine contained and managed imidacloprid-resistant Colorado potato beetles (Alyokhin et al., 2015).
Both IPM and One Health relegate environmentally damaging chemical controls to methods of last resort in part because of their repeated history of engendering resistance. However, the accelerating loss of pesticide and antibiotic efficacy testifies to the widespread appeal and dominance of chemical controls that persists despite the general understanding of their shortcomings. This is an important impediment to adopting more holistic, systemic, and sustainable approaches.
X. CHALLENGES
In its present form, resistance management faces significant headwinds. Diminishing new chemical options, combined with the persistent shortage of non-chemical alternatives, limits possibilities for their rotation or coadministration (Alyokhin et al., 2015). Environmental safety concerns complicate the development of new chemicals, genetic engineering, and release of new natural enemies. Such increased scrutiny reduces the risk of detrimental side effects but slows efforts to replace older agents lost to resistance.
We also face other socioeconomic challenges. The tragedy of the commons typifies resistance management, because short-term individual gain (often achieved by use of methods imposing strong selection) undermines a public good (the natural resource of long-term susceptibility) (Haywood et al., 2021; Mitchell & Onstad, 2023). Excessive market power, including monopoly and oligopoly, in the chemical and biotech industries stifles competition and innovation. Intellectual property laws intended to incentivize research and development can elevate the price of existing products (Slater, 2021). Conversely, loss of patent protection may undermine corporate incentives to manage resistance, especially once replacement products have come to market (Alyokhin et al., 2015). However, established manufacturing processes also provide a competitive advantage for original patent holders even after patent expirations.
To be successful, resistance management programs must address the needs and concerns of all stakeholders to the maximum possible extent (Gould et al., 2018; Hu, 2020; Shaw et al., 2023). The stakeholders, in turn, need to be well informed on all aspects of the problem and to be willing to compromise for the common good over longer timelines. Unfortunately, this is not always the case (Hu, 2020).
Industrialized farming, which is dominated by longstanding chemical and biotech customers, relies largely on pampered monocultures that meet market demands at the expense of increased susceptibility to pests and pathogens. Moreover, modern crop cultivars have been often bred under pathogen- and insect-protected conditions (often inadvertently eliminating innate resistance from the germplasm), assuming that such unchallenging conditions will also exist in the field due to widespread application of pesticides (Grant, 2007). Genetically diverse populations comprising different varieties and crosses, on the other hand, often perform better under biotic and abiotic stress than do genetically uniform monocultures (Zhu et al., 2000; Weedon et al., 2023).
Tailoring solutions to each problem (whether vaccines, narrow-spectrum biorational pesticides, or host-specific biological control agents) entails additional expense. This disincentivizes their use, especially when resources are scarce (Hurley & Frisvold, 2016). For example, extensive surveys studying decision-making by soybean, cotton, corn, and rice farmers in the USA indicated that costs were the main impediment to adopting herbicide resistance management practices (Weirich et al., 2011; Riar et al., 2013). Although this expense may prove justified by providing long-term sustainability and safety (Culliney, 2005; Rodrigues & Plotkin, 2020; Wyckhuys et al., 2020), short-term considerations often take precedence for enterprises concerned about their economic bottom line.
Chemical overuse may be exacerbated by the ‘moral hazard’ that exists when people and organizations in charge of providing information to end users, such as farmers, patients, and pest control operators, benefit from supplying more pesticides and drugs. Physicians employed by the clinics that directly sold drugs to their patients were more likely to prescribe antibiotics (Currie, Lin & Meng, 2014; Filippini, Heimsch & Masiero, 2014). Similarly, farmers who got their information on pest management from pesticide retailers were more likely to apply excessive amounts of chemicals, compared to farmers who received information from extension personnel (Jin, Bluemling & Mol, 2015).
Individual actors may also be reluctant to change their practices to address resistance management if their neighbours continue with the status quo (Gould et al., 2018; Haywood et al., 2021). In such a situation, resistance is likely to evolve in the immediate vicinity, and then spread through the dispersal of resistant organisms. Thus, the implemented resistance management measures will fail anyway, while the costs of their implementation, borne only by adopters, will be wasted.
Public interests, perceptions, markets, and political judgments often sacrifice resistance management (and other tactics with the promise to enhance long-term productivity and sustainability) in pursuit of therapeutic expediency, lower food prices, and higher agricultural profits. Resistance management may be low among competing priorities. Its importance may be underappreciated by decision makers, including many users of pesticides and antibiotics, due to lack of education about evolution and ecology. Eradication mentality, even when totally counterproductive, offers the false hope that easy and cheap solutions can overcome such problems. Unfortunately, initial success can never be sustained through such an approach (Dentzman, 2022). In addition, control alternatives, such as vaccines or transgenic plants, face public scepticism regardless of their scientific underpinning and demonstrated merits (Harmsen et al., 2013; Vega Rodríguez et al., 2022).
XI. PERSPECTIVES AND FUTURE DIRECTIONS
Though challenging, successful resistance management is essential to promote continued human progress and environmental sustainability. Widespread malnutrition and food insecurity undermine human development. Global warming, droughts and floods exacerbate pest and disease problems and facilitate invasions of non-native pests mediated by movement of people and cargo (Baker, Venugopal & Lamp, 2015; Yan et al., 2017; Pu et al., 2020). Habitat disturbance decreases ecosystem resistance and resilience to pest outbreaks. Depleting non-renewable resources demands increased productivity on the land and with the water that remains.
New technologies offer tools for managing resistant pests. For example, advances in molecular biology enhance the value of genetic engineering, which enables pesticides employing RNA interference (Mishra & Jurat-Fuentes, 2022), vaccines based on messenger RNA (mRNA) (Chen, Chen & Xu, 2022), and enhancing gene drives for pest control (Brossard et al., 2019). Precision application technologies employing remote sensing, robotics, and artificial intelligence enable targeted pest control with fewer side effects (Shafi et al., 2019; Marković et al., 2021). Increased computing power improves outbreak prediction, reducing ill-timed interventions (Marković et al., 2021; Li et al., 2021; Kishi et al., 2023).
Up until now only one or two plant protection genes have been deployed in any crop plant: tolerance for a single herbicide and one or two insect toxins. Existing technologies enable insertion into crop defensive signalling pathways of genes coding for diverse chemicals with highly distinct modes of action, mimicking the diverse onslaught of vertebrate immune responses. Induced expression promises less collateral damage to natural enemies and beneficials than does constitutive expression or systemic application. Diversifying the proteinase inhibitor pathways induced by chewing insects counters tolerance evolved to specific enzymes (Dunse et al., 2010), as does expressing components of pyrethrin in tomato trichomes (Wang et al., 2022). Expressing hemipteran-specific toxins in phloem-promotor regions targets piercing–sucking insects (McLean, 1998), and transforming crop plants to release certain volatiles, renders them less attractive to pests and more attractive to natural enemies (Turlings & Ton, 2006; Kos et al., 2013).
History, however, cautions against naïve faith in technological solutions (Huesemann & Huesemann, 2011). Resistance will evolve to new methods, just as it evolved to chemicals in the past. Sustainable management requires that we fully recognize our permanent evolutionary and ecological relationship with pathogens and pests and start acting accordingly. Complex problems require multiple, flexible solutions developed with thoughtfulness and humility. ‘Silver bullets’ are elusive not merely because they are silver (capable of solving a problem, once and for all), but also because they are ‘bullets’ (attempts at eradication, instead of systems that manage sustainable co-existence).
An alternative, but not mutually exclusive, approach involves enhancing the complexity, diversity, and dynamism of the potentially affected systems, better resembling natural and traditionally managed, pre-industrial environments. This may create an array of interconnections and feedback loops that lead to a greater ability to self-regulate, thus increasing their long-term stability (Nicholls & Altiery, 2007). For example, genetic diversification of a potato crop increased the sensitivity of the late blight pathogen Phytophthora infestans to commonly used fungicides mancozeb and azoxystrobin. Moreover, it significantly reduced genetic variation within the P. infestans population that could otherwise have served as a basis for the evolution of fungicide resistance (Yang et al., 2021).
At the risk of cliché, we need a paradigm shift. Pest and pathogen management systems must not merely reduce damage by economically feasible means; they must moreover display flexibility and the ability to adapt to changing circumstances (Fig. 1). Vaccines, less prone to resistance failure (Kennedy & Read, 2017, 2018), prime their host’s dynamic immune system, essentially enhancing an established co-evolutionary process. Biological control may do the same, but on larger spatial and temporal scales. Natural enemies are living organisms that are capable of evolutionary adaptations to changing biotic and abiotic conditions, including coevolution with their target pests (Wright & Bennett, 2018). They also may have dynamic numerical and behavioural responses to other members of an ecosystem that affect their abundance in a density-dependent manner (Onstad & Flexner, 2023). Other control methods are more static; nevertheless, inventing new ones in response to resistance can mimic a co-evolutionary process (Goeschl & Swanson, 2002; Alyokhin et al., 2015; Jørgensen et al., 2020).
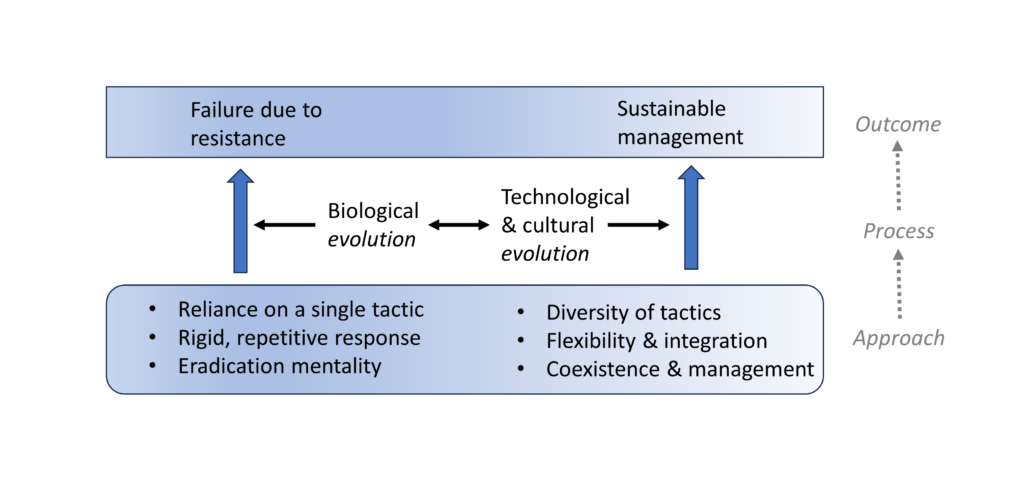
Immune responses are dynamic and diverse. Pathogens contend with nearly infinite defence modifications, whereas agricultural pests typically contend with only dozens of active ingredients. Similarly, pest management systems diverse in control methods and environmental conditions often benefit from greater stability, as do natural ecosystems characterized by high biodiversity (Loreau & de Mazancourt, 2013).
We generally lack the means to mount such timely and diverse responses through the invention of new control methods. Our technological tactics, while necessary, prove inadequate and unsustainable if deployed with ‘a total war psychology’. Leveraging existing, evolved resources (such as natural enemies, immune responses, and ecological resilience) as allies in a fully integrated approach holds more promise. Strong selection imposed by a single intervention puts us on a treadmill that an honest assessment of our capabilities would judge unsustainable. Only by wisely deploying and evoking the existing biological resources can we aspire to sustainable management strategies.
Managing resistance, an evolutionary process, demands a foundation in evolutionary biology. Developing sustainable solutions to resistance requires working with evolved immune and ecological systems, not against these evolutionary forces. We may never fully recreate the multimodal depth of an immune response when managing landscapes for human needs. Ecological systems are, after all, extremely complex, our knowledge is limited, and so are the resources available to improve this knowledge (Way & van Emden, 2000; Alyokhin & Kryukov, 2022). However, the features of immunity that prove highly resilient to dynamic challenges provide key insights that can and should be leveraged for more sustainable and profitable approaches to limiting the harms induced by pests and pathogens. By entraining resilient and varied responses, the benefits of vaccination generally endure. Diverse and redundant ecological systems capable of impeding the growth, replication, and dissemination of damaging organisms hold greater promise than the next powerful (but quickly undermined) chemical.
Multifaceted resistance problems also demand bridges among varied fields of science, avoiding redundancies and harnessing the efforts of theoreticians and practitioners. Successful resistance management requires transdisciplinary approaches, ranging from molecular genetics to philosophy, to tackle common problems.
XII. CONCLUSIONS
(1) Pests and pathogens develop resistance through evolutionary processes, especially when subjected to strong selection from one or just a few compounds or control measures.
(2) Human welfare and environmental preservation demand more sustainable approaches. These require management systems instead of individual management techniques.
(3) Such systems must be modelled on natural processes and be diverse and flexible enough to adapt to changing pest pressures.
(4) The foundation of sustainability for pest and pathogen management lies in true integration of diverse complementary control methods, natural host immunity, and coevolutionary ecological interactions.
XIII. ACKNOWLEDGEMENTS
This work is supported in part by the Multistate project award no. ME0-32125 from the U.S. Department of Agriculture’s National Institute of Food and Agriculture and by the US National Science Foundation One Health and the Environment (OH&E): Convergence of Social and Biological Sciences NRT program grant DGE-1922560. Any opinions, findings, conclusions, or recommendations expressed in this publication are those of the author(s) and should not be construed to represent any official USDA or U.S. Government determination or policy.
XIV. REFERENCES
Aaby, P., Netea, M.G. & Benn, C.S. (2023). Beneficial non-specific effects of live vaccines against COVID-19 and other unrelated infections. The Lancet Infectious Diseases 23, e34–e42.
Abedi, Z.H. & Brown, A.W.A. (1960). Development and reversion of DDT-resistance in Aedes aegypti. Canadian Journal of Genetics and Cytology 2, 252–261.
Adam, M., Murali, B., Glenn, N.O. & Potter, S.S. (2008). Epigenetic inheritance based evolution of antibiotic resistance in bacteria. BMC Evolutionary Biology 8, 52.
Adebisi, Y.A. (2023). Balancing the risks and benefits of antibiotic use in a globalized world: the ethics of antimicrobial resistance. Globalization and Health 19, 27.
Allen, C.T. (2008). Boll weevil eradication: an areawide pest management effort. In Areawide pest management: Theory and implementation (eds O. Koul, G. Cuperus & N. Elliott), pp. 467–559. CABI, Cambridge, MA.
Alves, J.M., Carneiro, M., Cheng, J.Y., de Matos, A.L., Rahman, M.M., Loog, L., Campos, P.F., Wales, N., Eriksson, A., Manica, A., Strive, T., Graham, S.C., Afonso, S., Bell, D.J., Belmont, L., et al. (2019). Parallel adaptation of rabbit populations to myxoma virus. Science 363, 1319–1326.
Alyokhin, A. & Chen, Y.H. (2017). Adaptation to toxic hosts as a factor in the evolution of insecticide resistance. Current Opinion in Insect Science 21, 33–38.
Alyokhin, A., Chen, Y.H., Udalov, M., Benkovskaya, G. & Lindström, L. (2022). Chapter 24 - Evolutionary considerations in potato pest management. In Insect Pests of Potato (Second Edition) (eds A. Alyokhin, S.I. Rondon & Y. Gao), pp. 429–450. Academic Press, Oxford, UK.
Alyokhin, A. & Kryukov, V. (2022). Chapter 25 - Ecology of a potato field. In Insect Pests of Potato (Second Edition) (eds A. Alyokhin, S.I. Rondon & Y. Gao), pp. 451–462. Academic Press, Oxford, UK.
Alyokhin, A., Mota-Sanchez, D., Baker, M., Snyder, W.E., Menasha, S., Whalon, M., Dively, G. & Moarsi, W.F. (2015). The Red Queen in a potato field: integrated pest management versus chemical dependency in Colorado potato beetle control. Pest Management Science 71, 343–356.
Andow, D.A. & Alstad, D.N. (1998). F2 screen for rare resistance alleles. Journal of Economic Entomology 91, 572–578.
Asser-Kaiser, S., Fritsch, E., Undorf-Spahn, K., Kienzle, J., Eberle, K.E., Gund, N.A., Reineke, A., Zebitz, C.P.W., Heckel, D.G., Huber, J. & Jehle, J.A. (2007). Rapid emergence of baculovirus resistance in codling moth due to dominant, sex-linked inheritance. Science 317, 1916–1918.
Bagavathiannan, M.V. & Davis, A.S. (2018). An ecological perspective on managing weeds during the great selection for herbicide resistance. Pest Management Science 74, 2277–2286.
Baker, M.B., Venugopal, P.D. & Lamp, W.O. (2015). Climate change and phenology: Empoasca fabae (Hemiptera: Cicadellidae) migration and severity of impact. PLoS ONE 10, e0124915.
Bang, C. & Schmitz, R.A. (2015). Archaea associated with human surfaces: not to be underestimated. FEMS Microbiology Reviews 39, 631–648.
Bass, C. & Field, L. (2011). Gene amplification and insecticide resistance. Pest Management Science 67, 886–890.
Bäumler, A.J. (2024). The coming microbial crisis: Our antibiotic bubble is about to burst. Science 385, eads3473.
Beckie, H.J., Busi, R., Lopez-Ruiz, F.J. & Umina, P.A. (2021). Herbicide resistance management strategies: how do they compare with those for insecticides, fungicides and antibiotics? Pest Management Science 77, 3049–3056.
Billman, G.E. (2020). Homeostasis: The underappreciated and far too often ignored central organizing principle of physiology. Frontiers in Physiology 11, 200.
Bobadilla, L.K. & Tranel, P.J. (2024). Predicting the unpredictable: the regulatory nature and promiscuity of herbicide cross resistance. Pest Management Science 80, 235–244.
Booy, O., Robertson, P.A., Moore, N., Ward, J., Roy, H.E., Adriaens, T., Shaw, R., Van Valkenburg, J., Wyn, G., Bertolino, S., Blight, O., Branquart, E., Brundu, G., Caffrey, J., Capizzi, D., et al. (2020). Using structured eradication feasibility assessment to prioritize the management of new and emerging invasive alien species in Europe. Global Change Biology 26, 6235–6250.
Bosch, R.V.D. (1978). The pesticide conspiracy. University of California Press, Berkeley.
Brevik, K., Bueno, E.M., McKay, S., Schoville, S.D. & Chen, Y.H. (2021). Insecticide exposure affects intergenerational patterns of DNA methylation in the Colorado potato beetle, Leptinotarsa decemlineata. Evolutionary Applications 14, 746–757.
Brossard, D., Belluck, P., Gould, F. & Wirz, C.D. (2019). Promises and perils of gene drives: navigating the communication of complex, post-normal science. Proceedings of the National Academy of Sciences 116, 7692–7697.
Burckhardt, R.M., Dennehy, J.J., Poon, L.L.M., Saif, L.J. & Enquist, L.W. (2022). Are COVID-19 vaccine boosters needed? The science behind boosters. Journal of Virology 96, e01973-21.
Caniça, M., Manageiro, V., Jones-Dias, D., Clemente, L., Gomes-Neves, E., Poeta, P., Dias, E. & Ferreira, E. (2015). Current perspectives on the dynamics of antibiotic resistance in different reservoirs. Research in Microbiology 166, 594–600.
Carabelli, A.M., Peacock, T.P., Thorne, L.G., Harvey, W.T., Hughes, J., de Silva, T.I., Peacock, S.J., Barclay, W.S., de Silva, T.I., Towers, G.J. & Robertson, D.L. (2023). SARS-CoV-2 variant biology: immune escape, transmission and fitness. Nature Reviews Microbiology 21, 162–177.
Carlson, J.M., Le, A.Q., Shahid, A. & Brumme, Z.L. (2015). HIV-1 adaptation to HLA: a window into virus–host immune interactions. Trends in Microbiology 23, 212–224.
Chait, R., Vetsigian, K. & Kishony, R. (2012). What counters antibiotic resistance in nature? Nature Chemical Biology 8, 2–5.
Chang, Z., Yadav, V., Lee, S.C. & Heitman, J. (2019). Epigenetic mechanisms of drug resistance in fungi. Fungal Genetics and Biology 132, 103253.
Chen, J., Chen, J. & Xu, Q. (2022). Current developments and challenges of mRNA vaccines. Annual Review of Biomedical Engineering 24, 85–109.
Chi, X., Li, Y. & Qiu, X. (2020). V(D)J recombination, somatic hypermutation and class switch recombination of immunoglobulins: mechanism and regulation. Immunology 160, 233–247.
Clemens, J., Brenner, R., Rao, M., Tafari, N. & Lowe, C. (1996). Evaluating new vaccines for developing countries - efficacy or effectiveness? Journal of the American Medical Association 275, 390–397.
Clements, J., Schoville, S., Peterson, N., Lan, Q. & Groves, R.L. (2016). Characterizing molecular mechanisms of imidacloprid resistance in select populations of Leptinotarsa decemlineata in the Central Sands region of Wisconsin. PLoS ONE 11, e0147844.
Coia, J.E., Duckworth, G.J., Edwards, D.I., Farrington, M., Fry, C., Humphreys, H., Mallaghan, C. & Tucker, D.R. (2006). Guidelines for the control and prevention of meticillin-resistant Staphylococcus aureus (MRSA) in healthcare facilities. Journal of Hospital Infection 63, S1–S44.
Conway, G.R. (1993). sustainable Agriculture: the trade-offs between with productivity, stability, and equitability. In Economics and Ecology. pp. 46–63. Chapman and Hall, London, UK.
Cooper, J. & Dobson, H. (2007). The benefits of pesticides to mankind and the environment. Crop Protection 26, 1337–1348.
Coughlan, L., Kremer, E.J. & Shayakhmetov, D.M. (2022). Adenovirus-based vaccines—a platform for pandemic preparedness against emerging viral pathogens. Molecular Therapy 30, 1822–1849.
Culliney, T.W. (2005). Benefits of classical biological control for managing invasive plants. Critical Reviews in Plant Sciences 24, 131–150.
Currie, J., Lin, W. & Meng, J. (2014). Addressing antibiotic abuse in China: An experimental audit study. Journal of Development Economics 110, 39–51.
Dang, K., Doggett, S., Singham, G. & Lee, C. (2017). Insecticide resistance and resistance mechanisms in bed bugs, Cimex spp. (Hemiptera: Cimicidae). Parasites & Vectors 10, 318.
Davies, J. & Davies, D. (2010). Origins and evolution of antibiotic resistance. Microbiology and Molecular Biology Reviews 74, 417–433.
Davis, A.S. & Frisvold, G.B. (2017). Are herbicides a once in a century method of weed control? Pest Management Science 73, 2209–2220.
Davis, A.S., Hill, J.D., Chase, C.A., Johanns, A.M. & Liebman, M. (2012). Increasing cropping system diversity balances productivity, profitability and environmental health. PLOS ONE 7, e47149.
Day, T., Kennedy, D.A., Read, A.F. & Gandon, S. (2022). Pathogen evolution during vaccination campaigns. PLoS Biology 20, e3001804.
Deb, B., Vilvadrinath, R. & Goel, S. (2021). COVID-19 variants that escape vaccine immunity: Global and Indian context-are more vaccines needed? Journal of Biosciences 46, 112.
Délye, C., Jasieniuk, M. & Corre, V.L. (2013). Deciphering the evolution of herbicide resistance in weeds. Trends in Genetics 29, 649–658.
Dentzman, K. (2022). Academics and the ‘easy button’: lessons from pesticide resistance management. Agriculture and Human Values 39, 1179–1183.
Dermauw, W. & Van Leeuwen, T. (2014). The ABC gene family in arthropods: comparative genomics and role in insecticide transport and resistance. Insect Biochemistry and Molecular Biology 45, 89–110.
Dimitriu, T., Matthews, A.C. & Buckling, A. (2021). Increased copy number couples the evolution of plasmid horizontal transmission and plasmid-encoded antibiotic resistance. Proceedings of the National Academy of Sciences 118, e2107818118.
Doole, G.J. & James, T.K. (2023). Profitable management of a finite herbicide resource. Crop Protection 172, 106314.
Dunse, K., Stevens, J., Lay, F., Gaspar, Y., Heath, R. & Anderson, M. (2010). Coexpression of potato type I and II proteinase inhibitors gives cotton plants protection against insect damage in the field. Proceedings of the National Academy of Sciences 107, 15011–15015.
Eckburg, P.B., Lepp, P.W. & Relman, D.A. (2003). Archaea and their potential role in human disease. Infection and Immunity 71, 591–596.
Ehrlich, P.R. & Raven, P.H. (1964). Butterflies and plants: a study in coevolution. Evolution 18, 586–608.
Faust, R.M. (2008). General introduction to areawide pest management. In Areawide pest management: theory and implementation (eds O. Koul, G. Cuperus & N. Elliott), pp. 1–14. CABI, Cambridge, MA.
Feyereisen, R., Dermauw, W. & Van Leeuwen, T. (2015). Genotype to phenotype, the molecular and physiological dimensions of resistance in arthropods. Pesticide Biochemistry and Physiology 121, 61–77.
Field, L.M., Devonshire, A.L., Ffrench-Constant, R.H. & Forde, B.G. (1989). Changes in DNA methylation are associated with loss of insecticide resistance in the peach-potato aphid Myzus persicae (Sulz.). FEBS Letters 243, 323–327.
Filippini, M., Heimsch, F. & Masiero, G. (2014). Antibiotic consumption and the role of dispensing physicians. Regional Science and Urban Economics 49, 242–251.
Fishbein, S.R.S., Mahmud, B. & Dantas, G. (2023). Antibiotic perturbations to the gut microbiome. Nature Reviews Microbiology, 1–17.
Flasche, S. & Atkins, K.E. (2018). Balancing benefits and risks of antibiotic use. The Journal of Infectious Diseases 218, 1351–1353.
Freeman, J.C., Smith, L.B., Silva, J.J., Fan, Y., Sun, H. & Scott, J.G. (2021). Fitness studies of insecticide resistant strains: lessons learned and future directions. Pest Management Science 77, 3847–3856.
Gage, K.L. & Schwartz-Lazaro, L.M. (2019). Shifting the paradigm: an ecological systems approach to weed management. Agriculture 9, 179.
Gatehouse, J.A. (2013). Genetic Engineering of Crops for Insect Resistance. In Sustainable Food Production (eds P. Christou, R. Savin, B.A. Costa-Pierce, I. Misztal & C.B.A. Whitelaw), pp. 808–845. Springer New York, New York, NY.
Geier, P.W., Clark, L.R. & Briese, D.T. (1983). Principles for the control of arthropod pests. Protection Ecology 5, 1–96.
Ghosh, D., Veeraraghavan, B., Elangovan, R. & Vivekanandan, P. (2020). Antibiotic resistance and epigenetics: more to it than meets the eye. Antimicrobial Agents and Chemotherapy 64, 10.1128/aac.02225-19.
Gillespie, D.R., Quiring, D.J.M., Foottit, R.G., Foster, S.P. & Acheampong, S. (2009). Implications of phenotypic variation of Myzus persicae (Hemiptera: Aphididae) for biological control on greenhouse pepper plants. Journal of Applied Entomology 133, 505–511.
Goeschl, T. & Swanson, T. (2002). On the economic limits to technological potential: Will industry resolve the resistance problem? In The Economics of Managing Biotechnologies (ed T. Swanson), pp. 99–128. Springer Netherlands, Dordrecht.
Goldman, E. (2021). How the unvaccinated threaten the vaccinated for COVID-19: A Darwinian perspective. Proceedings of the National Academy of Sciences 118.
Gould, F. (1998). Sustainability of transgenic insecticidal cultivars: Integrating pest genetics and ecology. Annual Review of Entomology 43, 701–726.
Gould, F., Brown, Z.S. & Kuzma, J. (2018). Wicked evolution: can we address the sociobiological dilemma of pesticide resistance? Science 360, 728–732.
Grant, S.M. (2007). The importance of biodiversity in crop sustainability: a look at monoculture. Journal of Hunger & Environmental Nutrition 1, 101–109.
Gray, S., De Boer, S., Lorenzen, J., Karasev, A., Whitworth, J., Nolte, P., Singh, R., Boucher, A. & Xu, H. (2010). Potato virus Y: an evolving concern for potato crops in the United States and Canada. Plant Disease 94, 1384–1397.
Green, S.J. & Grosholz, E.D. (2021). Functional eradication as a framework for invasive species control. Frontiers in Ecology and the Environment 19, 98–107.
Guerrero-Bosagna, C. & Skinner, M.K. (2012). Environmentally induced epigenetic transgenerational inheritance of phenotype and disease. Molecular and Cellular Endocrinology 354, 3–8.
Harmsen, I.A., Mollema, L., Ruiter, R.A., Paulussen, T.G., de Melker, H.E. & Kok, G. (2013). Why parents refuse childhood vaccination: a qualitative study using online focus groups. BMC Public Health 13, 1183.
Haywood, J., Vadlamani, G., Stubbs, K.A. & Mylne, J.S. (2021). Antibiotic resistance lessons for the herbicide resistance crisis. Pest Management Science 77, 3807–3814.
He, Z., Cheng, X., Kyzas, G.Z. & Fu, J. (2016). Pharmaceuticals pollution of aquaculture and its management in China. Journal of Molecular Liquids 223, 781–789.
Heap, I. (2023). The International Herbicide-Resistant Weed Database. www.weedscience.org [accessed 30 November 2023].
Henderson, P.J.F., Maher, C., Elbourne, L.D.H., Eijkelkamp, B.A., Paulsen, I.T. & Hassan, K.A. (2021). Physiological functions of bacterial “multidrug” efflux pumps. Chemical Reviews 121, 5417–5478.
Hernando-Amado, S., Coque, T.M., Baquero, F. & Martínez, J.L. (2019). Defining and combating antibiotic resistance from one health and global health perspectives. Nature Microbiology 4, 1432–1442.
Herzog, J., Müller, C.B. & Vorburger, C. (2007). Strong parasitoid-mediated selection in experimental populations of aphids. Biology Letters 3, 667–669.
Higley, L.G. & Pedigo, L.P. (1993). Economic injury level concepts and their use in sustaining environmental quality. Agriculture, Ecosystems & Environment 46, 233–243.
Hogan, M.J. & Pardi, N. (2022). mRNA vaccines in the COVID-19 pandemic and beyond. Annual Review of Medicine 73, 17–39.
Holman, D.B., Bearson, S.M.D., Bearson, B.L. & Brunelle, B.W. (2018). Chlortetracycline and florfenicol induce expression of genes associated with pathogenicity in multidrug-resistant Salmonella enterica serovar Typhimurium. Gut Pathogens 10, 10.
Hu, Z. (2020). What Socio-Economic and Political Factors Lead to Global Pesticide Dependence? A Critical Review from a Social Science Perspective. International Journal of Environmental Research and Public Health 17, 8119. Multidisciplinary Digital Publishing Institute.
Huang, F., Andow, D.A. & Buschman, L.L. (2011). Success of the high-dose/refuge resistance management strategy after 15 years of Bt crop use in North America. Entomologia Experimentalis et Applicata 140, 1–16.
Huesemann, M. & Huesemann, J. (2011). Techno-Fix: Why Technology Won’t Save Us Or the Environment. New Society Publishers, Gabriola Island, Canada.
Hueth, D. & Regev, U. (1974). Optimal agricultural pest management with increasing pest resistance. American Journal of Agricultural Economics 56, 543–552.
Hurley, T.M. & Frisvold, G. (2016). Economic Barriers to Herbicide-Resistance Management. Weed Science 64, 585–594.
Jhunjhunwala, S., Hammer, C. & Delamarre, L. (2021). Antigen presentation in cancer: insights into tumour immunogenicity and immune evasion. Nature Reviews Cancer 21, 298–312.
Jin, S., Bluemling, B. & Mol, A.P.J. (2015). Information, trust and pesticide overuse: Interactions between retailers and cotton farmers in China. NJAS: Wageningen Journal of Life Sciences 72–73, 23–32. Taylor & Francis.
Joglekar, A.V. & Li, G. (2021). T cell antigen discovery. Nature Methods 18, 873–880.
Jørgensen, P.S., Folke, C., Henriksson, P.J.G., Malmros, K., Troell, M. & Zorzet, A. (2020). Coevolutionary governance of antibiotic and pesticide resistance. Trends in Ecology & Evolution 35, 484–494.
Kennedy, D.A. & Read, A.F. (2017). Why does drug resistance readily evolve but vaccine resistance does not? Proceedings of the Royal Society B: Biological Sciences 284, 20162562.
Kennedy, D.A. & Read, A.F. (2018). Why the evolution of vaccine resistance is less of a concern than the evolution of drug resistance. Proceedings of the National Academy of Sciences 115, 12878–12886.
Kim, H., Choi, H.-G. & Shin, S.J. (2023). Bridging the gaps to overcome major hurdles in the development of next-generation tuberculosis vaccines. Frontiers in Immunology 14, 1193058.
Kishi, S., Sun, J., Kawaguchi, A., Ochi, S., Yoshida, M. & Yamanaka, T. (2023). Characteristic features of statistical models and machine learning methods derived from pest and disease monitoring datasets. Royal Society Open Science 10, 230079.
Knight, K.M., Head, G.P. & Rogers, D.J. (2021). Successful development and implementation of a practical proactive resistance management plan for Bt cotton in Australia. Pest Management Science 77, 4262–4273.
Kogan, M. & Jepson, P. (2007). Ecology, sustainable development, and IPM: The human factor. In Perspectives in Ecological Theory and Integrated Pest Management pp. 1–44. Cambridge University Press, Cambridge, UK.
Kos, M., Houshyani, B., Overeem, A., Bouwmeester, H., Weldegergis, B., van Loon, J., Dicke, M. & Vet, L. (2013). Genetic engineering of plant volatile terpenoids: effects on a herbivore, a predator and a parasitoid. Pest Management Science 69, 302–311.
Krell, T. & Matilla, M.A. (2022). Antimicrobial resistance: progress and challenges in antibiotic discovery and anti-infective therapy. Microbial Biotechnology 15, 70–78.
Labbé, P., Milesi, P., Yébakima, A., Pasteur, N., Weill, M. & Lenormand, T. (2014). Gene-dosage effects on fitness in recent adaptive duplications: ACE-1 in the mosquito Culex pipiens. Evolution 68, 2092–2101.
Laidlaw, B.J. & Ellebedy, A.H. (2022). The germinal centre B cell response to SARS-CoV-2. Nature Reviews Immunology 22, 7–18.
Laine, A.-L. (2009). Role of coevolution in generating biological diversity: spatially divergent selection trajectories. Journal of Experimental Botany 60, 2957–2970.
Leathwick, D.M., Ganesh, S. & Waghorn, T.S. (2015). Evidence for reversion towards anthelmintic susceptibility in Teladorsagia circumcincta in response to resistance management programmes. International Journal for Parasitology: Drugs and Drug Resistance 5, 9–15.
Levine, E., Oloumi-Sadeghi, H. & Fisher, J.R. (1992). Discovery of multiyear diapause in Illinois and South Dakota northern corn rootworm (Coleoptera: Chrysomelidae) eggs and incidence of the prolonged diapause trait in Illinois. Journal of Economic Entomology 85, 262–267.
Levine, E., Spencer, J.L., Isard, S.A., Onstad, D.W. & Gray, M.E. (2002). Adaptation of the western corn rootworm to crop rotation: evolution of a new strain in response to a management practice. American Entomologist 48, 94–107.
Li, X., Zhang, Z., Liang, B., Ye, F. & Gong, W. (2021). A review: antimicrobial resistance data mining models and prediction methods study for pathogenic bacteria. The Journal of Antibiotics 74, 838–849. Nature Publishing Group.
Lipsitch, M., Grad, Y.H., Sette, A. & Crotty, S. (2020). Cross-reactive memory T cells and herd immunity to SARS-CoV-2. Nature Reviews Immunology 20, 709–713.
Looft, T., Johnson, T.A., Allen, H.K., Bayles, D.O., Alt, D.P., Stedtfeld, R.D., Sul, W.J., Stedtfeld, T.M., Chai, B., Cole, J.R., Hashsham, S.A., Tiedje, J.M. & Stanton, T.B. (2012). In-feed antibiotic effects on the swine intestinal microbiome. Proceedings of the National Academy of Sciences 109, 1691–1696. Proceedings of the National Academy of Sciences.
Loreau, M. & de Mazancourt, C. (2013). Biodiversity and ecosystem stability: a synthesis of underlying mechanisms. Ecology Letters 16, 106–115.
Lu, K., Song, Y. & Zeng, R. (2021). The role of cytochrome P450-mediated detoxification in insect adaptation to xenobiotics. Current Opinion in Insect Science 43, 103–107.
Mackenzie, J.S. & Jeggo, M. (2019). The one health approach—why is it so important? Tropical Medicine and Infectious Disease 4, 88.
MacLean, R.C., Hall, A.R., Perron, G.G. & Buckling, A. (2010). The population genetics of antibiotic resistance: integrating molecular mechanisms and treatment contexts. Nature Reviews Genetics 11, 405–414.
Margaritopoulou, T., Tani, E., Chachalis, D. & Travlos, I. (2018). Involvement of epigenetic mechanisms in herbicide resistance: The case of Conyza canadensis. Agriculture 8, 17.
Marković, D., Vujičić, D., Tanasković, S., Đorđević, B., Ranđić, S. & Stamenković, Z. (2021). Prediction of pest insect appearance using sensors and machine learning. Sensors 21, 4846.
Markus, C., Pecinka, A., Karan, R., Barney, J.N. & Merotto Jr, A. (2018). Epigenetic regulation – contribution to herbicide resistance in weeds? Pest Management Science 74, 275–281.
Mason, P.G. (2021). Biological Control: Global Impacts, Challenges and Future Directions of Pest Management. CSIRO Publishing, Clayton South, Australia.
McLean, A. (1998). Vaccines and their impact on the control of disease. British Medical Bulletin 54, 545–556.
Meka, V.G., Gold, H.S., Cooke, A., Venkataraman, L., Eliopoulos, G.M., Moellering, R.C., Jr & Jenkins, S.G. (2004). Reversion to susceptibility in a linezolid-resistant clinical isolate of Staphylococcus aureus. Journal of Antimicrobial Chemotherapy 54, 818–820.
Meyer, H., Ehmann, R. & Smith, G.L. (2020). Smallpox in the post-eradication era. Viruses 12, 138.
Micoli, F., Bagnoli, F., Rappuoli, R. & Serruto, D. (2021). The role of vaccines in combatting antimicrobial resistance. Nature Reviews Microbiology 19, 287–302.
Mira-Martínez, S., Pickford, A.K., Rovira-Graells, N., Guetens, P., Tintó-Font, E., Cortés, A. & Rosanas-Urgell, A. (2019). Identification of antimalarial compounds that require CLAG3 for their uptake by plasmodium falciparum-infected erythrocytes. Antimicrobial Agents and Chemotherapy 63, 10.1128/aac.00052-19.
Mishra, S. & Jurat-Fuentes, J.L. (2022). Insecticidal RNA interference (RNAi) for control of potato pests. In Insect Pests of Potato (Second Edition) (eds A. Alyokhin, S.I. Rondon & Y. Gao), pp. 219–229. Academic Press, Oxford, UK.
Mitchell, P.D. & Onstad, D.W. (2023). Valuing pest susceptibility to control. In Insect Resistance Management (Third Edition) (eds D.W. Onstad & L.M. Knolhoff), pp. 31–59. Academic Press, London, UK.
Mogilicherla, K. & Roy, A. (2023). Epigenetic regulations as drivers of insecticide resistance and resilience to climate change in arthropod pests. Frontiers in Genetics 13. Frontiers.
Mortensen, D.A., Ryan, M.R. & Smith, R.G. (2024). Another step on the transgene-facilitated herbicide treadmill. Pest Management Science Early View.
Mota-Sanchez, D. & Wise, J.C. (2023). The Arthropod Pesticide Resistance Database. www.pesticideresistance.org [accessed 30 November 2023].
Muraduzzaman, A.K.M., Illing, P.T., Mifsud, N.A. & Purcell, A.W. (2022). Understanding the role of HLA class I molecules in the immune response to influenza infection and rational design of a peptide-based vaccine. Viruses 14, 2578.
Murray, C.J.L., Ikuta, K.S., Sharara, F., Swetschinski, L., Aguilar, G.R., Gray, A., Han, C., Bisignano, C., Rao, P., Wool, E., Johnson, S.C., Browne, A.J., Chipeta, M.G., Fell, F., Hackett, S., et al. (2022). Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. The Lancet 399, 629–655. Elsevier.
Nascimento, D.V., Lira, R. & Torres, J.B. (2023). Heterosis and reselection for pyrethroid resistance trait maintenance in the lady beetle Eriopis connexa (Germar). Annals of Applied Biology 183, 148–158.
National Research Council (1986). Pesticide resistance: Strategies and tactics for management. The National Academies Press, Washington, DC.
Nauen, R., Bass, C., Feyereisen, R. & Vontas, J. (2022). The role of cytochrome P450s in insect toxicology and resistance. Annual Review of Entomology 67, 105–124.
Nicholls, C.I. & Altiery, M.A. (2007). Agroecology: contributions toward a renewed ecological foundation for pest management. In Perspectivess in Ecological Theory and Integrated Pest Management pp. 431–468. Cambridge University Press, Cambridge, UK.
Nowak, M.A., May, R.M. & Anderson, R.M. (1990). The evolutionary dynamics of HIV-1 quasispecies and the development of immunodeficiency disease. AIDS (London, England) 4, 1095–1103.
Oakeshott, J., Farnsworth, C., East, P., Scott, C., Han, Y., Wu, Y. & Russell, R. (2013). How many genetic options for evolving insecticide resistance in heliothine and spodopteran pests? Pest Management Science 69, 889–896.
Ocan, M., Akena, D., Nsobya, S., Kamya, M.R., Senono, R., Kinengyere, A.A. & Obuku, E.A. (2019). Persistence of chloroquine resistance alleles in malaria endemic countries: a systematic review of burden and risk factors. Malaria Journal 18, 76.
Ojemaye, C.Y. & Petrik, L. (2019). Pharmaceuticals in the marine environment: a review. Environmental Reviews 27, 151–165.
Onstad, D.W. (2023). Modeling for prediction and management. In Insect Resistance Management (Third Edition) (eds D.W. Onstad & L.M. Knolhoff), pp. 401–433. Academic Press, London, UK.
Onstad, D.W. & Flexner, J.L. (2023). Insect resistance, natural enemies, and density-dependent processes. In Insect Resistance Management (Third Edition) (eds D.W. Onstad & L.M. Knolhoff), pp. 381–399. Academic Press, London, UK.
Onstad, D.W. & Knolhoff, L.M. (2023). Major issues in insect resistance management. In Insect Resistance Management (Third Edition) (eds D.W. Onstad & L.M. Knolhoff), pp. 1–29. Academic Press.
Oppold, A., Kreß, A., Vanden Bussche, J., Diogo, J.B., Kuch, U., Oehlmann, J., Vandegehuchte, M.B. & Müller, R. (2015). Epigenetic alterations and decreasing insecticide sensitivity of the Asian tiger mosquito Aedes albopictus. Ecotoxicology and Environmental Safety 122, 45–53.
Oppold, A.-M. & Müller, R. (2017). Chapter Nine - Epigenetics: A Hidden Target of Insecticides. In Insect Epigenetics (ed H. Verlinden), pp. 313–324. Academic Press.
Panini, M., Manicardi, G., Moores, G. & Mazzoni, E. (2016). An overview of the main pathways of metabolic resistance in insects. ISJ-Invertebrate Survival Journal 13, 326–335.
Patra, S., Raney, M., Pareek, A. & Kaur, R. (2022). Epigenetic Regulation of Antifungal Drug Resistance. Journal of Fungi 8, 875. Multidisciplinary Digital Publishing Institute.
Perotti, V.E., Larran, A.S., Palmieri, V.E., Martinatto, A.K. & Permingeat, H.R. (2020). Herbicide resistant weeds: A call to integrate conventional agricultural practices, molecular biology knowledge and new technologies. Plant Science 290, 110255.
Peterson, E. & Kaur, P. (2018). Antibiotic resistance mechanisms in bacteria: Relationships between resistance determinants of antibiotic producers, environmental bacteria, and clinical pathogens. Frontiers in Microbiology 9, 426686.
Pisa, L., Goulson, D., Yang, E.-C., Gibbons, D., Sánchez-Bayo, F., Mitchell, E., Aebi, A., van der Sluijs, J., MacQuarrie, C.J.K., Giorio, C., Long, E.Y., McField, M., Bijleveld van Lexmond, M. & Bonmatin, J.-M. (2021). An update of the Worldwide Integrated Assessment (WIA) on systemic insecticides. Part 2: impacts on organisms and ecosystems. Environmental Science and Pollution Research 28, 11749–11797.
Prokopy, R.J. (1993). Stepwise progress towards IPM and sustainable agriculture. IPM Practitioner 15, 1–4.
Pu, J., Wang, Z. & Chung, H. (2020). Climate change and the genetics of insecticide resistance. Pest Management Science 76, 846–852.
Quesada, H.B., Baptista, A.T.A., Cusioli, L.F., Seibert, D., de Oliveira Bezerra, C. & Bergamasco, R. (2019). Surface water pollution by pharmaceuticals and an alternative of removal by low-cost adsorbents: A review. Chemosphere 222, 766–780.
Radwan, J., Babik, W., Kaufman, J., Lenz, T.L. & Winternitz, J. (2020). Advances in the evolutionary understanding of MHS polymorphism. Trends in Genetics 36, 298–311.
Raszick, T.J. (2021). Boll weevil eradication: A success story of science in the service of policy and industry. Annals of the Entomological Society of America 114, 702–708.
Raymond, B. (2019). Five rules for resistance management in the antibiotic apocalypse, a road map for integrated microbial management. Evolutionary Applications 12, 1079–1091.
Riar, D.S., Norsworthy, J.K., Steckel, L.E., Iv, D.O.S., Eubank, T.W., Bond, J. & Scott, R.C. (2013). Adoption of Best Management Practices for Herbicide-Resistant Weeds in Midsouthern United States Cotton, Rice, and Soybean. Weed Technology 27, 788–797.
Robotham, J.V., Deeny, S.R., Fuller, C., Hopkins, S., Cookson, B. & Stone, S. (2016). Cost-effectiveness of national mandatory screening of all admissions to English National Health Service hospitals for meticillin-resistant Staphylococcus aureus: a mathematical modelling study. The Lancet Infectious Diseases 16, 348–356. Elsevier.
Rodrigues, C.M.C. & Plotkin, S.A. (2020). Impact of vaccines: Health, economic and social perspectives. Frontiers in Microbiology 11, 1526.
Roth, G.A., Picece, V.C.T.M., Ou, B.S., Luo, W., Pulendran, B. & Appel, E.A. (2022). Designing spatial and temporal control of vaccine responses. Nature Reviews Materials 7, 174–195.
Roush, R.T. & McKenzie, J.A. (1987). Ecological genetics of insecticide and acaricide resistance. Annual Review of Entomology 32, 361–380.
Sah, P., Medlock, J., Fitzpatrick, M.C., Singer, B.H. & Galvani, A.P. (2018). Optimizing the impact of low-efficacy influenza vaccines. Proceedings of the National Academy of Sciences 115, 5151–5156.
Salyers, A.A. & Amábile-Cuevas, C.F. (1997). Why are antibiotic resistance genes so resistant to elimination? Antimicrobial Agents and Chemotherapy 41, 2321–2325.
Seeley, T. (2017). Darwinian beekeeping: an evolutionary approach to apiculture. American Bee Journal 157, 277–282.
Segrestin, J., Götzenberger, L., Valencia, E., de Bello, F. & Lepš, J. (2024). A unified framework for partitioning the drivers of stability of ecological communities. Global Ecology and Biogeography 33, e13828.
Shafi, U., Mumtaz, R., García-Nieto, J., Hassan, S.A., Zaidi, S.A.R. & Iqbal, N. (2019). Precision agriculture techniques and practices: From considerations to applications. Sensors 19, 3796.
Shalini, K., Anwer, Z., Sharma, P.K., Garg, V.K. & Kumar, N. (2010). A review on pharma pollution. International Journal of PharmTech Research 2, 2265–2270.
Sharma, P., Wollenberg, K., Sellers, M., Zainabadi, K., Galinsky, K., Moss, E., Nguitragool, W., Neafsey, D. & Desai, S.A. (2013). An epigenetic antimalarial resistance mechanism involving parasite genes linked to nutrient uptake. Journal of Biological Chemistry 288, 19429–19440.
Sharrock, J. & Sun, J.C. (2020). Innate immunological memory: from plants to animals. Current Opinion in Immunology 62, 69–78.
Shaw, D.R., Asmus, A.B., Schroeder, J. & Ervin, D.E. (2023). Changing the paradigm for pesticide resistance management. Pest Management Science 79, 4726–4730.
Sherrard-Smith, E., Skarp, J.E., Beale, A.D., Fornadel, C., Norris, L.C., Moore, S.J., Mihreteab, S., Charlwood, J.D., Bhatt, S., Winskill, P., Griffin, J.T. & Churcher, T.S. (2019). Mosquito feeding behavior and how it influences residual malaria transmission across Africa. Proceedings of the National Academy of Sciences 116, 15086–15095.
Shim, E. & Galvani, A. (2012). Distinguishing vaccine efficacy and effectiveness. Vaccine 30, 6700–6705.
Shukla, R., Ramasamy, V., Shanmugam, R.K., Ahuja, R. & Khanna, N. (2020). Antibody-dependent enhancement: a challenge for developing a safe dengue vaccine. Frontiers in Cellular and Infection Microbiology 10, 572681.
Simberloff, D. (2001). Eradication of island invasives: practical actions and results achieved. Trends in Ecology & Evolution 16, 273–274.
Singh, A. & Leppanen, C. (2020). Known target and nontarget effects of the novel neonicotinoid cycloxaprid to arthropods: A systematic review. Integrated Environmental Assessment and Management 16, 831–840.
Singh, V. & Chibale, K. (2021). Strategies to combat multi-drug resistance in tuberculosis. Accounts of Chemical Research 54, 2361–2376.
Slater, E. (2021). Poverty via monopolization: The impact that intellectual property rights and federal subsidies have on farm poverty. Journal of Intellectual Property Law 29, 209–231.
Spagnolo, F., Trujillo, M. & Dennehy, J. (2021). Why do antibiotics exist? MBIO 12, e01966-21.
Stiling, P. (2002). Ecology: theories and applications.Third. Prentice Hall, Upper Saddle River, NJ.
Suchan, T. & Alvarez, N. (2015). Fifty years after Ehrlich and Raven, is there support for plant–insect coevolution as a major driver of species diversification? Entomologia Experimentalis et Applicata 157, 98–112.
Suckling, D.M., Stringer, L.D., Stephens, A.E., Woods, B., Williams, D.G., Baker, G. & El-Sayed, A.M. (2014). From integrated pest management to integrated pest eradication: technologies and future needs. Pest Management Science 70, 179–189.
Sullivan, J.C., De Meyer, S., Bartels, D.J., Dierynck, I., Zhang, E.Z., Spanks, J., Tigges, A.M., Ghys, A., Dorrian, J., Adda, N., Martin, E.C., Beumont, M., Jacobson, I.M., Sherman, K.E., Zeuzem, S., et al. (2013). Evolution of treatment-emergent resistant variants in telaprevir phase 3 clinical trials. Clinical Infectious Diseases 57, 221–229.
Susser, S., Vermehren, J., Forestier, N., Welker, M.W., Grigorian, N., Füller, C., Perner, D., Zeuzem, S. & Sarrazin, C. (2011). Analysis of long-term persistence of resistance mutations within the hepatitis C virus NS3 protease after treatment with telaprevir or boceprevir. Journal of Clinical Virology 52, 321–327.
Tabashnik, B.E. (1994). Evolution of resistance to Bacillus thuringiensis. Annual Review of Entomology 39, 47–79.
Tabashnik, B.E., Brevault, T. & Carriere, Y. (2013). Insect resistance to Bt crops: lessons from the first billion acres. Nature Biotechnology 31, 510–521.
Tabashnik, B.E., Fabrick, J.A. & Carrière, Y. (2023). Global patterns of insect resistance to transgenic Bt crops: The first 25 years. Journal of Economic Entomology 116, 297–309.
Tabashnik, B.E., Mota-Sanchez, D., Whalon, M.E., Hollingworth, R.M. & Carriere, Y. (2014). Defining terms for proactive management of resistance to Bt crops and pesticides. Journal of Economic Entomology 107, 496–507.
Tabashnik, B.E., Sisterson, M.S., Ellsworth, P.C., Dennehy, T.J., Antilla, L., Liesner, L., Whitlow, M., Staten, R.T., Fabrick, J.A., Unnithan, G.C., Yelich, A.J., Ellers-Kirk, C., Harpold, V.S., Li, X. & Carrière, Y. (2010). Suppressing resistance to Bt cotton with sterile insect releases. Nature Biotechnology 28, 1304–1307.
Tedijanto, C., Olesen, S.W., Grad, Y.H. & Lipsitch, M. (2018). Estimating the proportion of bystander selection for antibiotic resistance among potentially pathogenic bacterial flora. Proceedings of the National Academy of Sciences 115, E11988–E11995.
The Brundtland Commission (1987). Report of the world commission on environment and development: our common future. In p. 300. The United Nations, New York, NY.
Tranel, P.J. (2021). Herbicide resistance in Amaranthus tuberculatus. Pest Management Science 77, 43–54.
Tu, Y. & Rappel, W.-J. (2018). Adaptation in living systems. Annual Review of Condensed Matter Physics 9, 183–205.
Turlings, T. & Ton, J. (2006). Exploiting scents of distress: the prospect of manipulating herbivore-induced plant odours to enhance the control of agricultural pests. Current Opinion in Plant Biology 9, 421–427.
Unno, T., Han, D., Jang, J., Lee, S.-N., Kim, J.H., Ko, G., Kim, B.G., Ahn, J.-H., Kanaly, R.A., Sadowsky, M.J. & Hur, H.-G. (2010). High diversity and abundance of antibiotic-resistant Escherichia coli isolated from humans and farm animal hosts in Jeonnam Province, South Korea. Science of The Total Environment 408, 3499–3506.
Van Meerbeek, K., Jucker, T. & Svenning, J.-C. (2021). Unifying the concepts of stability and resilience in ecology. Journal of Ecology 109, 3114–3132.
Vandermeer, J. & Andow, D.A. (1986). Prophylactic and responsive components of an integrated pest management program. Journal of Economic Entomology 79, 299–302.
Vega Rodríguez, A., Rodríguez-Oramas, C., Sanjuán Velázquez, E., Hardisson de la Torre, A., Rubio Armendáriz, C. & Carrascosa Iruzubieta, C. (2022). Myths and realities about genetically modified food: a risk-benefit analysis. Applied Sciences 12, 2861.
Wang, Y., Wen, J., Liu, L., Chen, J., Wang, C., Li, Z., Wang, G., Pichersky, E. & Xu, H. (2022). Engineering of tomato type VI glandular trichomes for trans-chrysanthemic acid biosynthesis, the acid moiety of natural pyrethrin insecticides. Metabolic Engineering 72, 188–199.
Way, M.J. & van Emden, H.F. (2000). Integrated pest management in practice — pathways towards successful application. Crop Protection 19, 81–103.
Weedon, O.D., Brumlop, S., Haak, A., Baresel, J.P., Borgen, A., Döring, T., Goldringer, I., Lammerts van Bueren, E., Messmer, M.M., Mikó, P., Nuijten, E., Pearce, B., Wolfe, M. & Finckh, M.R. (2023). High buffering potential of winter wheat composite cross populations to rapidly changing environmental conditions. Agronomy 13, 1662.
Wei, C.-J., Crank, M.C., Shiver, J., Graham, B.S., Mascola, J.R. & Nabel, G.J. (2020). Next-generation influenza vaccines: opportunities and challenges. Nature Reviews Drug Discovery 19, 239–252.
Weirich, J.W., Shaw, D.R., Owen, M.D., Dixon, P.M., Weller, S.C., Young, B.G., Wilson, R.G. & Jordan, D.L. (2011). Benchmark study on glyphosate-resistant cropping systems in the United States. Part 5: Effects of glyphosate-based weed management programs on farm-level profitability. Pest Management Science 67, 781–784.
Welch, T. & LaPatra, S. (2016). Yersinia ruckeri lipopolysaccharide is necessary and sufficient for eliciting a protective immune response in rainbow trout (Oncorhynchus mykiss Walbaum). Fish & Shellfish Immunology 49, 420–426.
Wright, M.G. & Bennett, G.M. (2018). Evolution of biological control agents following introduction to new environments. BioControl 63, 105–116.
Wyckhuys, K.A.G., Sasiprapa, W., Taekul, C. & Kondo, T. (2020). Unsung heroes: Fixing multifaceted sustainability challenges through insect biological control. Current Opinion in Insect Science 40, 77–84.
Yan, Y., Wang, Y.-C., Feng, C.-C., Wan, P.-H.M. & Chang, K.T.-T. (2017). Potential distributional changes of invasive crop pest species associated with global climate change. Applied Geography 82, 83–92.
Yang, L.-N., Nkurikiyimfura, O., Pan, Z.-C., Wang, Y.-P., Waheed, A., Chen, R.-S., Burdon, J.J., Sui, Q.-J. & Zhan, J. (2021). Plant diversity ameliorates the evolutionary development of fungicide resistance in an agricultural ecosystem. Journal of Applied Ecology 58, 2566–2578.
Ye, C.-Y., Tang, W., Wu, D., Jia, L., Qiu, J., Chen, M., Mao, L., Lin, F., Xu, H., Yu, X., Lu, Y., Wang, Y., Olsen, K.M., Timko, M.P. & Fan, L. (2019). Genomic evidence of human selection on Vavilovian mimicry. Nature Ecology & Evolution 3, 1474–1482.
Yewdell, J.W. & Santos, J.J.S. (2021). Original antigenic sin: how original? How sinful? Cold Spring Harbor Perspectives in Medicine 11, a038786.
Zalom, F.G. (1993). Reorganizing to facilitate the development and use of integrated pest management. Agriculture, Ecosystems & Environment 46, 245–256.
Zhu, Y., Chen, H., Fan, J., Wang, Y., Li, Y., Chen, J., Fan, J., Yang, S., Hu, L., Leung, H., Mew, T.W., Teng, P.S., Wang, Z. & Mundt, C.C. (2000). Genetic diversity and disease control in rice. Nature 406, 718–722. Nature Publishing Group.
Zimmermann, P. & Curtis, N. (2019). Factors that influence the immune response to vaccination. Clinical Microbiology Reviews 32, e00084-18.
Zost, S.J., Parkhouse, K., Gumina, M.E., Kim, K., Diaz Perez, S., Wilson, P.C., Treanor, J.J., Sant, A.J., Cobey, S. & Hensley, S.E. (2017). Contemporary H3N2 influenza viruses have a glycosylation site that alters binding of antibodies elicited by egg-adapted vaccine strains. Proceedings of the National Academy of Sciences 114, 12578–12583.